Bench to Bedside Webinar Focuses on COVID-19: Vaccine and Testing Challenges
The latest webinar in the Bench to Bedside series on June 30 addressed the state of COVID-19 testing and vaccine development.
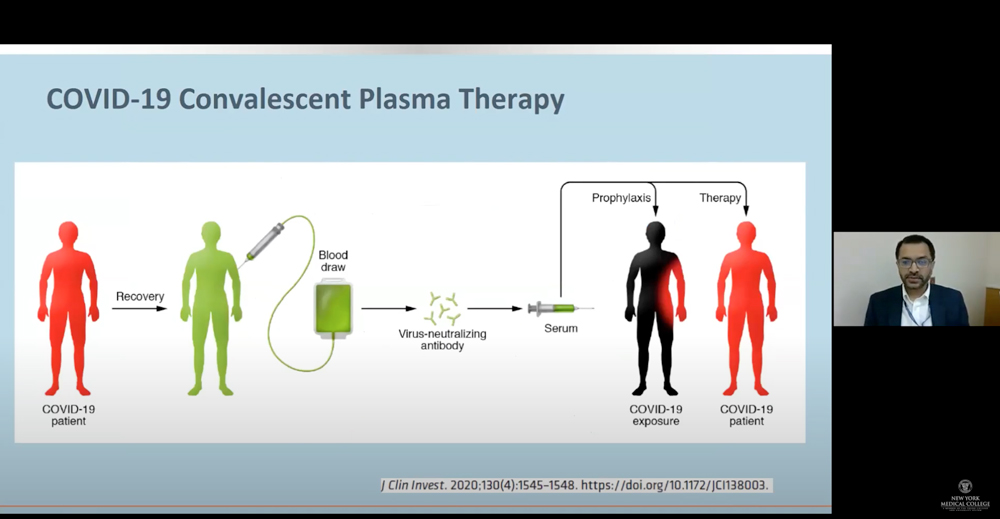
Moderated by Salomon Amar, D.D.S., Ph.D., vice president for research at New York Medical College (NYMC) and provost for biomedical research at Touro College and University System (TCUS), the webinar featured four experts who covered approaches, implementation, interpretation and clinical implications of COVID-19 testing, testing critically ill patients and vaccine and antiviral drug development. “In any crisis, you need to look from a brighter perspective. To see all the segments of the biomedical industry, whether in academia, hospitals or research institutions, coming together and working together for the same goal to develop novel strategies to be able to ward off the pandemic, should be commended,” said Dr. Amar.
The first speaker, Donald Chen, M.D., assistant professor of medicine and hospital epidemiologist and medical director of infection prevention at Westchester Medical Center (WMC), who is a co-principle investigator of the collaborative clinical drug trial by NYMC and Regeneron Pharmaceuticals which was launched at WMC, with the potential to treat critically ill patients with COVID-19, presented “COVID-19 Testing Approaches and Implementation.” Dr. Chen covered the hospitals’ strategy for whom to test and which test to use, how to optimize true positive and true negative results, types of commercial tests available and their limitations and the role of antibody testing.
Lawrence DeLorenzo, M.D., professor of clinical medicine and director, Medical Intensive Care Unit, WMC, who is also a co-principle investigator of the collaborative clinical drug trial with NYMC, Regeneron and WMC, presented “Testing of the Critically Ill COVID Patient in the Intensive Care Unit.” He spoke on hospital considerations of the virus including emergency room protocols, clinical care challenges in the ICU, protection of staff, non-COVID patients and visitors and hospital discharges. Dr. DeLorenzo stressed the importance of the ability to be flexible which included the capability of expanding from 11 ICU beds to 80 ICU beds and the significance of imaging including chest x-rays, CT scans, ultrasound and MRIs.
Next up was Abhay Dhand, M.D., associate professor of medicine and director, Transplant Infectious Diseases, WMC, who presented “Interpretation and Clinical Implications of COVID-19 Testing.” He described the basic mechanisms of the disease from incubation to illness onset, diagnostic methods, the issue of the variance of viral loads by sample type and disease stage and implications of antigen testing. Dr. Dhand also explained the limited understanding and complexities of the magnitude and duration of antibody responses in persons with different disease severity, genetics, comorbidities, age and/or infection history.
Finally, Robert W. Rubin, Ph.D., president and chief executive officer, Lovelace Biomedical Research Institute, provided an entrepreneurial perspective. Dr. Rubin presented “Drug Development in a Time of Crisis: Preclinical Approaches to Emergency Vaccine and Antiviral Drug Development. Experience with SARS-CoV-II.” He described why drug development takes so long and common stumbling blocks, accelerated approaches for antiviral and vaccine development, preclinical testing and the Food and Drug Administration approval process. Dr. Rubin concluded that it will take a variety of approaches and multiple vaccines and antiviral treatments to stem the COVID-19 pandemic.
